AbbVie Announces New Formulary Listings for SKYRIZI® in the Treatment of Moderate to Severe Plaque Psoriasis
- SKYRIZI® is an interleukin-23 (IL-23) inhibitor used for the treatment of moderate to severe psoriasis.1
- In clinical studies, SKYRIZI® significantly improved levels of skin clearance after just 16 weeks and maintained clearance at one year (52 weeks).1
- Non-Insured Health Benefits program lists SKYRIZI® on its formulary effective February 21, 2020.
- Manitoba lists SKYRIZI® on its provincial formulary effective March 02, 2020.
- SKYRIZI® is the only IL-23 inhibitor to arrive at a positive conclusion with the pCPA and is currently listed on the formularies of Alberta, Saskatchewan, Manitoba, Ontario, Quebec, Nova Scotia and the Non-Insured Health Benefits program.
MONTRÉAL, March 11, 2020 /CNW/ - AbbVie (NYSE: ABBV), a global, research and development-based biopharmaceutical company, announced today that SKYRIZI® (risankizumab) is now listed on the formularies of the Non-Insured Health Benefits program (NIHB) and the province of Manitoba for the treatment of moderate to severe plaque psoriasis. This announcement comes just three months after AbbVie successfully reached an agreement with the pan-Canadian Pharmaceutical Alliance (pCPA), bringing a much-needed treatment to patients living with psoriasis.
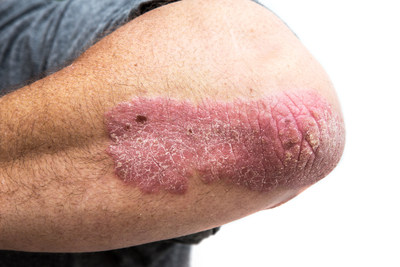
Psoriasis is a chronic condition affecting 125 million people worldwide, including 1 million Canadians, and many patients despite treatment still do not reach their goals or lose treatment response over time.2-4 The most common form is plaque psoriasis, which affects approximately 90% of patients. 5
SKYRIZI® (risankizumab) is part of a collaboration between Boehringer Ingelheim and AbbVie, with AbbVie leading development and commercialization globally.
"It's been very rewarding to see my psoriasis patients on the biologic SKYRIZI® respond so favorably. In my patients, I witness both a durable skin response and an improvement in quality of life and well-being," states Dr. Marni C. Wiseman MD FRCPC, Dermatologist from SKiNWISE DERMATOLOGY. "There currently is a need for treatments with longer dosing intervals for patients located in remote communities. I am so pleased that Manitobans and Canada's Indigenous population will have better access to this treatment," adds Dr. Wiseman.
According to the 2016 census, Aboriginal peoples in Canada totaled 1,673,785 people, or 4.9% of the national population.6 Of these, 223,310 Aboriginal people live in Manitoba, making up 18.0% of that population.7
For the Non-Insured Health Benefits program (NIHB), SKYRIZI® is effectively listed on NIHB's Limited Use formulary as of February 21, 2020 and criteria details will be featured in their next bulletin update.
Information regarding the Manitoba provincial criteria may be found by consulting the following link:
About SKYRIZI®
SKYRIZI® is a novel, humanized immunoglobulin monoclonal antibody designed to selectively inhibit IL-23, a naturally occurring cytokine involved in inflammatory and immune responses.1 On April 17, 2019, SKYRIZI™ received a NOC from Health Canada for the treatment of moderate to severe plaque psoriasis in patients who are candidates for systemic therapy or phototherapy, based on results from clinical studies showing significant improvement in levels of skin clearance after just 16 weeks and at 52 weeks with every 3-month dosing in more than 2000 adult patients.1 Four pivotal Phase 3 studies, ultIMMa-1, ultIMMa-2, IMMvent and IMMhance evaluated more than 2,000 patients with moderate to severe plaque psoriasis.4 Canadians living with moderate to severe plaque psoriasis were well represented in all four of the pivotal clinical trials leading to Health Canada's approval, showing the Canadian leadership in this clinical development program.
About AbbVie Care
The AbbVie Care program is designed to provide a wide range of customized services including reimbursement and financial support, pharmacy services, lab work reminders and coordination, personalized education and ongoing disease management support throughout the treatment journey. For more information, consult www.abbviecare.ca.
About AbbVie
AbbVie is a global, research and development-based biopharmaceutical company committed to developing innovative advanced therapies for some of the world's most complex and critical conditions. The company's mission is to use its expertise, dedicated people and unique approach to innovation to markedly improve treatments across four primary therapeutic areas: immunology, oncology, virology and neuroscience. In more than 75 countries, AbbVie employees are working every day to advance health solutions for people around the world. For more information about AbbVie, please visit us at www.abbvie.ca and www.abbvie.com. Follow @abbvieCanada and @abbvie on Twitter or view careers on our Facebook or LinkedIn page.
References:
| 1. | SKYRIZI® (risankizumab) [Canadian Product Monograph]. AbbVie Corporation, April 17, 2019. |
| 2. | International Federation of Psoriasis Associations. Accessed March 22, 2019. Available at: https://ifpa-pso.com/wp-content/uploads/2017/01/Brochure-Psoriasis-is-a-serious-disease-deserving-global-attention.pdf. |
| 3. | Mroweitz, U., et al. Definition of treatment goals for moderate to severe psoriasis: a European consensus. Arch Dermatol Res. 2011 Jan; 303(1): 1–10. |
| 4. | Levin, et al. Biologic fatigue in psoriasis. J Dermatolog Treat. 2014 Feb;25(1):78-82. doi: 10.3109/09546634.2013.826341. |
| 5. | Canadian Dermatology Association – Psoriasis. Accessed on February 5, 2020. |
| 6. | Statistics Canada, Aboriginal Population Profile, 2016 Census. |
| 7. | Statistics Canada, Focus on Geography Series, Province of Manitoba, 2016 Census. |

SOURCE AbbVie
