Opportunity for biotech firms to pursue drug candidates that target metabolic disorders, says GlobalData
Gene therapies have been a point of discussion during the last several years as a potential curative option for a variety of disease indications. The majority of the cardiovascular and metabolic disorder (CVMD) gene therapies in development are targeting cardiovascular indications, indicating immense opportunity for biotech companies to pursue drug candidates that target metabolic disorders, says leading data and analytics company GlobalData.
The company’s report, ‘Gene Therapy in CVMD’ focused on CVMD gene therapies, which included candidates targeting coronary artery disease, critical limb ischemia, diabetic foot ulcers and Pompe Disease, and that are in development in the eight major markets (8MM; the US, France, Germany, Italy, Spain, the UK, Japan and urban China). The report found that big pharma companies lag behind small bio-techs in R&D efforts.
In addition, the report reveals that, despite investment in CVMD gene therapies, the majority of the candidates across the 8MM are still in early R&D stages, whereas only a handful of CVMD gene therapies are in Phase II or Phase III clinical trials.
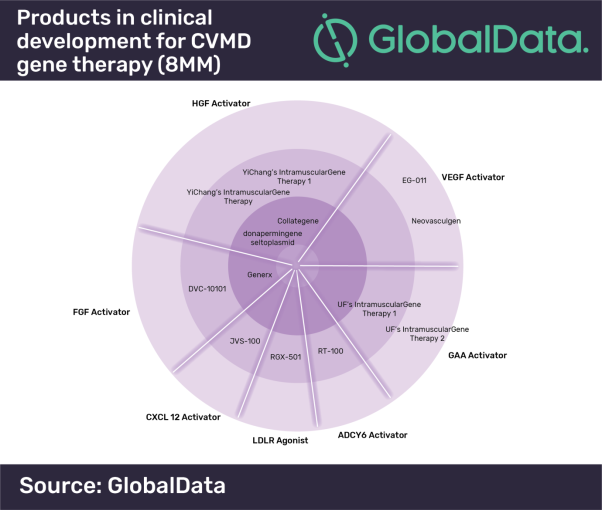
Within the entire CVMD space, the current gene therapy pipeline features approximately 13 candidates that are in Phase I, II or III of clinical development. Only three gene therapies have reached Phase III trials: AnGes MG’s Collategene, Angionetics’ Generx and ViroMed’s donaperminogene seltoplasmid.
Pavan Kottamasu, Healthcare Analyst at GlobalData, says: “Of the pipeline CVMD gene therapies, the majority of them target cardiovascular indications such as coronary artery disease, peripheral artery disease, critical limb ischemia, and systolic heart failure, indicating a massive opportunity for biotech companies to develop drug candidates that target metabolic disorders.”
Key opinion leaders interviewed by GlobalData anticipate big pharma companies to partner with these smaller biotech companies to enhance development of gene therapy candidates within the next decade.
