AbbVie reaches an agreement with the pan-Canadian Pharmaceutical Alliance (pCPA) for VENCLEXTA®, a treatment for chronic lymphocytic leukemia (CLL)
- Following the signing of an agreement between AbbVie and the pan-Canadian Pharmaceutical Alliance (pCPA), Ontario is the first province to reimburse VENCLEXTA.
- VENCLEXTA is the first and only BCL-2 inhibitor to be approved in Canada.
- A need exists for CLL patients who have failed novel oral therapies.i
MONTREAL, May 23, 2019 /CNW/ - AbbVie (NYSE: ABBV), a global, research and development-driven biopharmaceutical company, announced today that Ontario is the first province to reimburse VENCLEXTA® (venetoclax) monotherapy under its Drugs and Devices Division's (DDD) Exceptional Access Program effective May 13, 2019.ii VENCLEXTA is indicated for the treatment of patients with chronic lymphocytic leukemia (CLL) who have received at least one prior therapy and who have no other treatment options. VENCLEXTA is an oral, once-daily medicine.
This listing follows the signing of an agreement between AbbVie and the pan-Canadian Pharmaceutical Alliance (pCPA) earlier this year.
"People living with chronic lymphocytic leukemia in Ontario finally have access to VENCLEXTA, which is great news considering the nature of this disease. When faced with CLL, which can be recurrent, it is important to offer physicians and their patients highly effective options to tackle this illness," explains Elizabeth Lye, Director of Research & Programs at Lymphoma Canada. "At Lymphoma Canada, we hope that soon all Canadians will have access to this treatment."
VENCLEXTA monotherapy will be available through the Exceptional Access Program for the treatment of relapsed or refractory chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) in patients who have failed treatment with at least one prior therapy (including B-cell receptor inhibitors) or who have experienced intolerance to ibrutinib.
"When I meet a patient for the first time, I always explain that CLL is a type of blood cancer that is chronic and that some may relapse after being in remission. In fact, most of the patients I see in my clinic have often already gone through another line of treatment. Therefore, I want to ensure I have the appropriate treatments available for my patients in a timely manner," says Dr. Peter Anglin, MD, FRCPC, MBA, Hematologist and Physician Lead, Stronach Regional Cancer Centre. "I am thrilled that Ontario is the first province to reimburse venetoclax; it's a positive step for people living with CLL who need access to this much needed treatment."
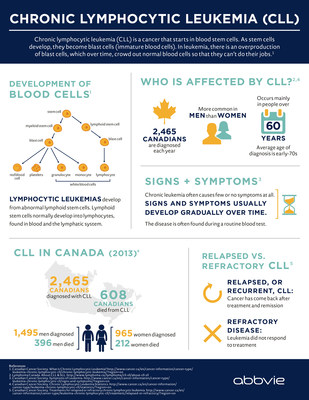
CLL, which is typically a slow-progressing cancer of the bone marrow and bloodiii, is one of the most common types of leukemia in adults. In Canada, CLL accounts for approximately 2,465 newly diagnosed cases of leukemia each year and is responsible for more than 600 deaths a year.iv
"It's been a long time coming for wonderful news like this. I can remember reading about clinical trials for this treatment several years ago and thinking how promising it sounded for people like me who have already been treated with more than one line of therapy and relapsed," states Mark Silverstein of Aurora, Ontario. "Now it's finally here! Another treatment option in the CLL world has been approved and reimbursed in the province of Ontario, and hopefully soon coming to every Canadian province. And thank you AbbVie for your commitment to bringing VENCLEXTA to those who need it."
VENCLEXTA received a Notice of Compliance with Conditions (NOC/c) from Health Canada on September 30, 2016. The therapy was approved as monotherapy for previously treated CLL patients, who have either a genetic mutation known as 17p deletion, or who do not have the mutation but have no other available treatment options, and who have received at least one other therapy. The 17p deletion is a genetic mutation that is found in 3 to 10 percent of people with previously untreated CLL and up to 50 percent of relapsed or refractory cases.v Under Health Canada's NOC/c policy, AbbVie will provide Health Canada with data from additional studies to assess the clinical benefit of VENCLEXTA in these patient populations.vi
"This agreement with the pCPA and subsequently the listing in Ontario are positive steps towards patients living with CLL accessing a medication that serves an unmet medical need," stated Stéphane Lassignardie, General Manager of AbbVie Canada. "AbbVie is committed to changing the way blood cancers are treated with innovative new treatment options."
VENCLEXTA continues to be investigated in CLL and other hematological diseases.
VENCLEXTA is being developed by AbbVie and Genentech, a member of the Roche Group. It is jointly commercialized by the companies in the U.S. and by AbbVie outside of the U.S.
About AbbVie Care
Canadians prescribed VENCLEXTA will have the opportunity to be enrolled in AbbVie Care, AbbVie's signature care program. The program is designed to provide a wide range of customized services including reimbursement and financial support, pharmacy services, lab work reminders and coordination, personalized education and ongoing disease management support throughout the treatment and beyond.
About AbbVie
AbbVie is a global, research and development-driven biopharmaceutical company committed to developing innovative advanced therapies for some of the world's most complex and critical conditions. The company's mission is to use its expertise, dedicated people and unique approach to innovation to markedly improve treatments across four primary therapeutic areas: immunology, oncology, virology and neuroscience. In more than 75 countries, AbbVie employees are working every day to advance health solutions for people around the world. For more information about AbbVie, please visit us at www.abbvie.ca and www.abbvie.com. Follow @abbvieCanada and @abbvie on Twitter or view careers on our Facebook or LinkedIn page.
| __________________________________ |
| iJain P. et al. Long-Term Outcomes for Patients With Chronic Lymphocytic Leukemia Who Discontinue IbrutinibCancer. 2017 Jun 15;123(12):2268-2273 |
| ii Ontario Ministry of Health and Long-Term Care http://health.gov.on.ca/en/pro/programs/drugs/eap_mn.aspx. Accessed May 2019 |
| iii Lymphoma Canada. Chronic lymphocytic leukemia. Available at http://www.lymphoma.ca/lymphoma/lymphoma-101/types-lymphoma/cll. Accessed May 2019 |
| iv Canadian Cancer Statistics. Chronic lymphocytic leukemia statistics http://www.cancer.ca/en/cancer-information/cancer-type/leukemia-chronic-lymphocytic-cll/statistics/?region=on. Accessed May 2019 |
| v Schnaiter A. et al. 17p deletion in chronic lymphocytic leukemia: risk stratification and therapeutic approachHematol Oncol Clin N Am. 2013;27:289–301 |
| vi VENCLEXTA product monograph, AbbVie Corporation. Date of Preparation: September 27, 2016. Date of Revision: February 12, 2019 http://www.abbvie.ca/content/dam/abbviecorp/ca/en/docs/VENCLEXTA_PM_EN.pdf. Accessed May 2019 |

