Baxter Canada Announces First Regulatory Approval of New Novum IQ Infusion Platform
- Pioneering platform offers both large volume and syringe infusion pumps, is foundational for continued innovation in medication delivery and management
- Integrates Baxter's decades of leadership in infusion therapy with advanced digital health solutions, including the Dose IQ Safety Software—a web-based drug library software—and the IQ Enterprise Connectivity Suite, to build on the company's proven technology for protecting infusions from medication error
- Offers both English and French language options to meet the needs of healthcare professionals across Canada
- Health Canada approval represents first regulatory clearance in a series of planned submissions globally
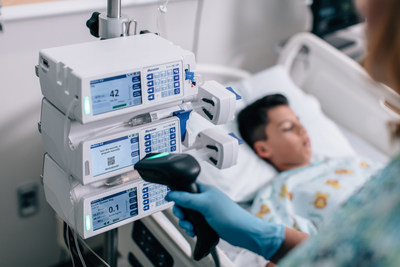
MISSISSAUGA, ON, Nov. 12, 2020 /CNW/ - Baxter Canada, a leader in innovative technology for medication delivery, today announced Health Canada marketing authorization of its new Novum IQ Infusion Platform. Building on the company's decades of experience at the patient bedside, Novum IQ is the foundation for Baxter's continued innovation in medication delivery and management. The marketing authorization in Canada includes Novum IQ large volume (LVP) and syringe infusion pumps as well as the company's Dose IQ Safety Software, representing Baxter's latest developments for infusion therapy and medication safety. Novum IQ is expected to be available to Canadian customers in the coming weeks.

"We are excited to introduce Novum IQ, our newest infusion pump platform designed to provide personalized therapy and help prevent harm to patients." said Anne-Marie Carli, head of Baxter's Medication Delivery business in Canada. "The integrated platform provides for both large volumetric and syringe infusion pumps adding another option for Canadian hospitals to provide high-quality infusion therapy for patients. Novum IQ is a full-suite package offered in both English and French."
Protecting More Infusions with Advanced Technologies
Advancing the safety and efficiency of medication delivery and management is critical, as data continues to show the harmful burden of errors on patient safety.1,2 In addition to offering both LVP and syringe infusion pumps, Novum IQ utilizes intuitive technologies built to protect infusions.
Novum IQ features Baxter's Dose IQ Safety Software, a web-based, customizable drug library and dose error reduction system. A well-managed drug library is a key safety feature of smart infusion pumps to assist clinicians in reducing the occurrence of potential dosing errors. Dose IQ helps hospitals by:
- Helping ensure pumps are up-to-date with the latest dose information through centralized access to drug library files
- Working across LVP and syringe pumps with the ability to differentiate enteral delivery on the syringe pump through designated screen colors
- Incorporating titration error prevention technology, providing additional safety measures for infusions
Novum IQ fully integrates with hospital electronic medical records (EMRs) through Baxter's IQ Enterprise Connectivity Suite – a software which also:
- Enables over-the-air drug library and software updates to all Novum IQ pumps connected to a hospital's network
- Provides clear visualizations of infusion data across a hospital system, allowing efficient identification of safety and process issues at every level of care
Based on clinical setting and patient need, hospitals benefit from having access to both LVP and syringe pumps. As LVPs deliver fluid from intravenous (IV) bags and syringe pumps from a syringe, LVPs typically administer higher volumes of fluid at faster rates. Syringe pumps are often used to precisely deliver small amounts of fluid at low rates, often in pediatric, neonatal or anesthesia care settings. The Novum IQ Infusion Platform integrates user experience across its LVP and syringe pumps, with shared safety features, advanced connectivity and a consistent, intuitive interface.
About Baxter Canada
Every day, millions of patients and caregivers rely on Baxter's leading portfolio of critical care, nutrition, renal, hospital and surgical products. In Canada, we've been operating at the critical intersection where innovations that save and sustain lives meet the healthcare providers that make it happen for 83 years. With products, technologies and therapies available in more than 100 countries, Baxter's employees worldwide are now building upon the company's rich heritage of medical breakthroughs to advance the next generation of transformative healthcare innovations. Baxter Canada and its over 1,200 employees are located primarily in Ontario at the Head Office, CIVA Admixing and Technical Services Centres in Mississauga, and in Alliston – where Baxter operates Canada's only large-scale manufacturing plant producing life-sustaining intravenous and dialysis solutions. To learn more, visit www.baxter.ca and follow Baxter on LinkedIn
For safe and proper use of this device, refer to the Operator Manual.
Indications for Use:
Novum IQ Large Volume Pump
The Novum IQ LVP is intended to be used for the controlled administration of fluids. These include parenteral pharmaceutical drugs, nutrition, blood and blood products.
The Novum IQ LVP is intended to deliver an infusion through clinically acceptable routes of administration: intravenous, arterial, subcutaneous, and epidural.
The Novum IQ LVP is intended to be used in conjunction with legally marketed and compatible intravenous administration sets, and medications provided by the user.
The Novum IQ LVP is suitable for patient care in hospitals and outpatient health care facilities.
The Novum IQ LVP is intended for use on adults, pediatrics and neonates.
The Novum IQ LVP is intended to aid in the reduction of operator interaction through guided programming, including a way to automate the programming of infusion parameters and documentation of infusion therapies. This automation is intended to aid in the reduction of programming errors.
The Novum IQ LVP is intended to be used by trained healthcare professionals.
Novum IQ Syringe Pump:
The Novum IQ Syringe Infusion Pump is intended to be used for the controlled administration of fluids. These include parenteral pharmaceutical drugs, nutrition, blood and blood products, and enteral nutrition.
The Novum IQ Syringe Infusion Pump is intended to deliver an infusion through clinically accepted routes of administration: intravenous, arterial, enteral, and subcutaneous.
The Novum IQ Syringe Infusion Pump is intended to be used in conjunction with legally marketed and compatible administration sets, syringes, and medications provided by the user.
The Novum IQ Syringe Infusion Pump is suitable for patient care in hospitals and outpatient health care facilities.
The Novum IQ Syringe Infusion Pump is intended for use on adults, pediatrics and neonates.
The Novum IQ Syringe Infusion Pump is intended to aid in the reduction of operator interaction through guided programming, including a way to automate the programming of infusion parameters and documentation of infusion therapies. This automation is intended to aid in the reduction of programming errors.
The Novum IQ Syringe Infusion Pump is intended to be used by trained healthcare professionals.
Baxter, Novum IQ, Dose IQ and IQ Enterprise are registered trademarks of Baxter International Inc.
| 1 | https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5770837/pdf/bmjopen-2017-015912.pdf |
| 2 | https://www.fda.gov/drugs/drug-information-consumers/working-reduce-medication-errors |
SOURCE Baxter Corporation
