Baxter Launches Prismax In Canada to Maximize Treatment in the Intensive Care Unit
- Next-generation platform for continuous renal replacement therapy is designed to simplify treatment delivery and accuracy
- Incorporates latest technology and innovative features inspired by clinician feedback
- TherMax blood warmer delivers effective blood warming for a variety of organ support therapies
MISSISSAUGA, ON, Aug. 12, 2019 /CNW/ - Baxter Canada, a leader in acute care, announced today Health Canada's approval of its PrisMaxsystem and integrated TherMaxblood warmer---the company's next-generation platform for continuous renal replacement therapy (CRRT), therapeutic plasma exchange (TPE) and organ support therapies (OST). The PrisMaxsystem was developed with input from more than 650 healthcare providers globally and is designed to help simplify the complexities of delivering CRRT while providing more time for patient care in the Intensive Care Unit (ICU).
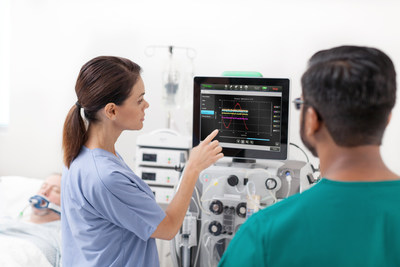
"We're excited to bring PrisMax to Canadian ICUs, where clinicians are often fighting the clock to meet the unique needs of their critically ill patients," said Victoria Jurincic, head of Baxter's renal business in Canada. "This system's innovative features help simplify therapy delivery and maximize its accuracy and efficiency. With 20 years of expertise in CRRT and blood filtering technology, Baxter is uniquely positioned to bring this platform to market and help our customers as they strive to save and sustain lives."
Building on Baxter's leading Prismaflextechnology currently used by hospitals in more than 90 countries, the PrisMaxsystem is used to treat patients with acute kidney injury (AKI)—a common complication in the ICU that is associated with a greater risk of adverse outcomes for critically ill patients1—and fluid overload. Of the more than 400,000 patients admitted to Canadian ICUs every year, approximately 25-30 percent will develop AKI during their hospitalization—an incidence rate that has been on the rise both in Canada and globally2. According to a study that examined Canadian critical care units, patients with AKI were more likely to die than those without it. 3
Baxter designed the PrisMaxsystem to optimize treatment accuracy and system performance, while making it simpler and more efficient for clinicians to use. Treatment accuracy is a critical component of delivering the benefits of CRRT, and the system uses intelligent pump and scale adjustments to help ensure that fluid removal targets prescribed by the patient's clinical team are met. PrisMaxalso offers clinicians flexibility for effluent management, with the option of auto-effluent drain or effluent bag drain configurations. Additionally, a prospective, multicenter, international pilot study found that PrisMax delivered significant improvements in areas that impact efficiency and ease of use, including the time needed for bag changes, the number of informational and malfunction alarms, how often the blood pump stops, filter life and machine downtime.4
The integrated TherMaxblood warmer, which is used exclusively with the PrisMax system, is an important component for extracorporeal therapies to warm the blood prior to returning to the body, helping keep the patient's body temperature at a normal level. TherMax also includes several advanced patient safety features to help control blood return temperature, detect leaks, and to ensure the correct setup.5
Baxter has launched PrisMaxin more than 20 countries across Europe, the United States and Australia, and expects to file for regulatory approval of PrisMaxin additional countries in 2019 and 2020.
About Baxter Canada
Every day, millions of patients and caregivers rely on Baxter's leading portfolio of critical care, nutrition, renal, hospital and surgical products. In Canada, we've been operating at the critical intersection where innovations that save and sustain lives meet the healthcare providers that make it happen for 82years. With products, technologies and therapies available in more than 100 countries, Baxter's employees worldwide are now building upon the company's rich heritage of medical breakthroughs to advance the next generation of transformative healthcare innovations. Baxter Canada and its nearly 1,000 employees are located primarily in Ontario at the Head Office, CIVA Admixing and Technical Services Centres in Mississauga, and in Alliston – where Baxter operates Canada's only large-scale manufacturing plant producing life-sustaining intravenous and dialysis solutions. To learn more, visit www.baxter.ca and follow us on Twitter, LinkedIn and Facebook
Rx Only.For safe and proper use of this device, refer to the full Instructions for Use.
This release includes forward-looking statements concerning PrisMax and TherMax, including potential benefits associated with their use. The statements are based on assumptions about many important factors, including the following, which could cause actual results to differ materially from those in the forward-looking statements: satisfaction of regulatory and other requirements; actions of regulatory bodies and other governmental authorities; product quality, manufacturing or supply, or patient safety issues; changes in law and regulations; and other risks identified in Baxter's most recent filing on Form 10-K and other SEC filings, all of which are available on Baxter's website. Baxter does not undertake to update its forward-looking statements.
Baxter, PrisMax, TherMax, and Prismaflex are registered trademarks of Baxter International Inc.
| 1 Odutayo Can J Anesth 2012 |
| 2 Wald-AJKD Incidence and Outcomes 2014 |
| 3 Can J Anesth/J Can Anesth (2012) 59:934–942 |
| 4 Broman M, et alBlood Purif. 2018;46:220-227 |
| 5 Baxter - TherMax Operators Manual. AW7006 Rev A 2018;Sep |

SOURCE Baxter Corporation
